GFAP Rabbit Polyclonal Antibody
| Cat Number: | AB-10682 |
|---|---|
| Conjugate: | Unconjugated |
| Size: | 100 ul |
| Clone: | POLY |
| Concentration: | 1mg/ml |
| Host: | Rabbit |
| Isotype: | IgG |
| Immunogen: | Purified bovine full length protein |
| Reactivity: | Hu, Ms, Rt, Ct, Mamm. |
| Applications: | Western Blot: 1: 2,500 is recommended |
| Molecular Weight: | 55kDa |
| Purification: | Serum |
| Background: | Glial Fibrillary Acidic Protein (GFAP) was discovered by Amico Bignami and coworkers as a majorfibrous protein of multiple sclerosis plaques (1). It was subsequently found to be a member of the 10nm or intermediate filament protein family, specifically the intermediate filament protein family Class III, which also includes peripherin, desmin and vimentin. The GFAP protein runs on gels at ~55kDa protein, usually associated with lower molecule weight bands which are thought to be proteolytic fragments and alternate transcripts from the single gene. GFAP is strongly and specifically expressed in astrocytes and certain other astroglia in the central nervous system, in satellite cells in peripheral ganglia, and in non-myelinating Schwann cells in peripheral nerves. In many damage and disease states GFAP expression is heavily upregulated in astrocytes. In addition neural stem cells frequently strongly express GFAP. Antibodies to GFAP are therefore very useful as markers of astrocytic cells and neural stem cells. In addition many types of brain tumor, presumably derived from astrocytic cells, heavily express GFAP. Finally, Alexander’s disease was recently shown to be caused by point mutations in protein coding region of the GFAP gene (2). All forms of Alexander disease are characterized by the presence of Rosenthal fibers, which are GFAP containing cytoplasmic inclusions found in astrocytes. Human, horse, cow, pig, chicken, rat, mouse and other mammalians. |
| Form: | Liquid |
| Buffer: | Antibody is supplied as an aliquot of serum plus 5mM NaN3 |
| Storage: | At 4°-8° C for short term. At -20°C For longer term. Avoid repeated freezing and thawing cycles. |
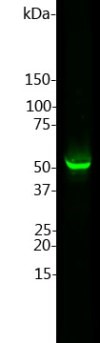
Left: Western blot of whole rat
cerebellum homogenate stained with GFAP, at dilution of 1:2,500. A prominent band running with an apparent SDS-PAGE molecular weight of ~50kDa corresponds to rodent
GFAP. A lower band at ~45kDa is derived from the GFAP molecule.
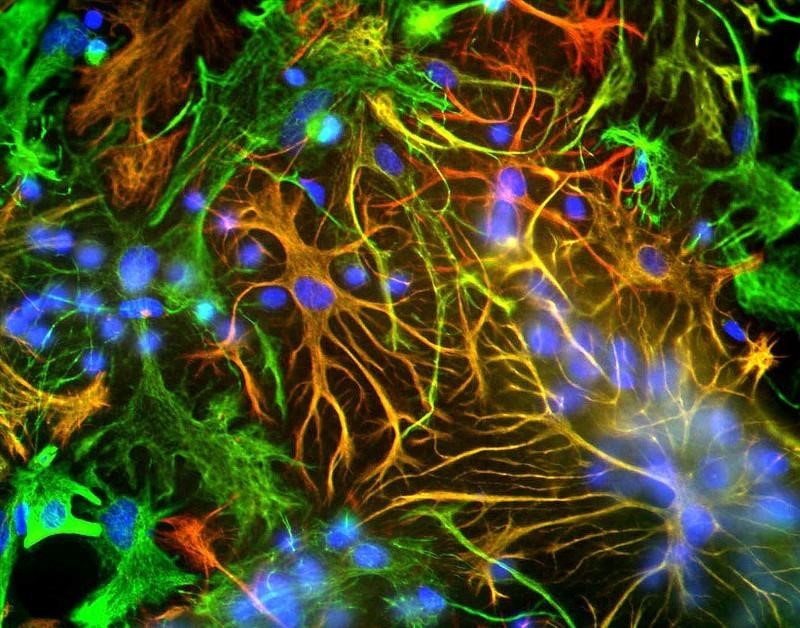
Immunocytochemistry/Immunofluorescence: GFAP Antibody ICC-IF analysis of mixed neuron-glial cultures using GFAP antibody (red) and Vimentin antibody (green). The fibroblastic cells contain only Vimentin and so are green. The astrocytes contain either Vimentin and GFAP (appearing golden) or predominantly GFAP (appearing red). Blue is nuclear DNA stain
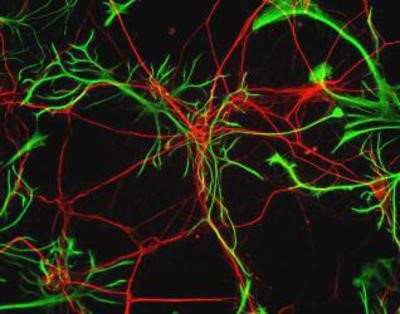
Immunocytochemistry/Immunofluorescence: GFAP Antibody – Rat neurons stained with Neurofilament Heavy antibody (red) and GFAP antibody (green).
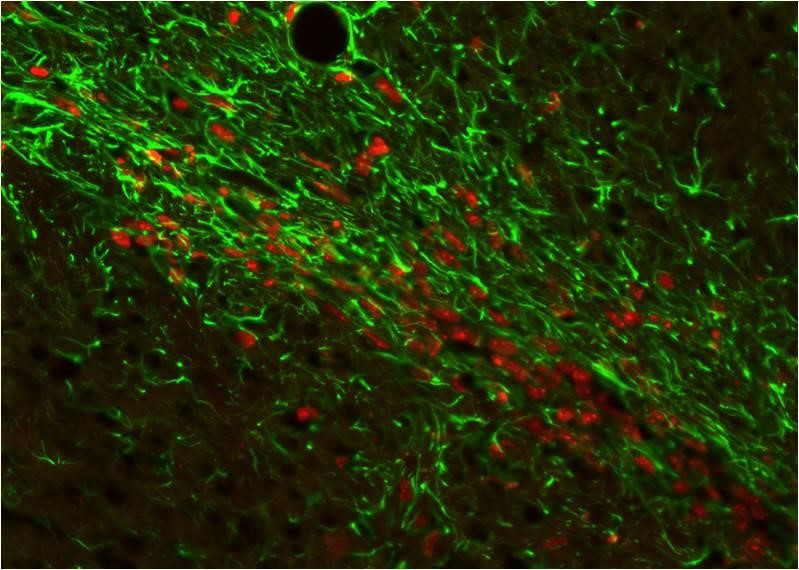
Immunocytochemistry/Immunofluorescence: GFAP Antibody Xenografted mouse brain section : astocyte and human nuclei.
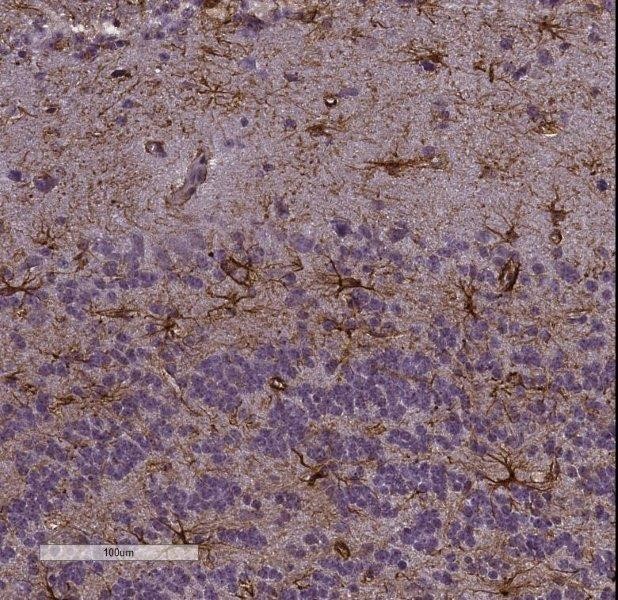
Immunohistochemistry-Frozen: GFAP Antibody Imaging of mouse brain (cortex), 20x magnification
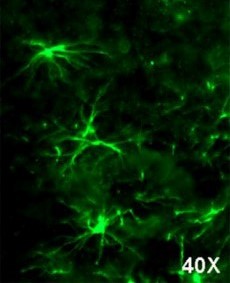
Immunohistochemistry frozen tissues At a 1/500 dilution staining rat spinal cord tissue sections by IHC-Fr. Rats were transcardially perfused with 4% PFA. The tissue was post fixed 1 hour in 4% PFA and then 30% sucrose for three days. 20µm sections were cryostat cut. The primary antibody was incubated with the tissue sections for 18 hours. Bound antibody was detected using an Alexa Fluor 488 conjugated goat anti-rabbit polyclonal
PROCEDURE OF IMMUNOFLUORESCENT STAINING OF FREE-FLOATING BRAIN TISSUE SECTIONSTISSUE PREPARATION:
1. Perfuse transcardially the animal (rat or mouse) with ice-cold PBS (pH7.4), followed by freshly made 4% paraformaldehyde fixative solution in PBS.
2. Postfix the removed brain in the same 4% paraformaldehyde fixative solution in PBS (4°C for 16 – 24 hours).
3. Cryoprotect the tissue by immersing it in sucrose solutions in PBS (15%, for 24 hours followed by 30% until tissue will sink, may take from 48 hours up to 1 week).
4. Cut 40 – 50μm sections on a cryostat.
5. Keep sections in PBS + 0.05M NaN3 at 4°C
until they were taken for staining. During the staining process, the sections should never be allowed to dry out.
